New Developments in Albatross Conservation—Using biologging to elucidate behavior in the ocean

Figure 1: A Laysan albatross with GPS, camera, and accelerometer data logger attached to its back.
Photo credit: Nishizawa Bungo

Watanuki Yutaka, Professor, School of Fisheries Science, Hokkaido University
The albatross is a group of birds at high risk of extinction. The reasons for this are thought to include a decreased survival rate of parent birds due to bycatching during fishing. To date, the research of incidental by-catch risk and the evaluation of the efficiency of by-catch mitigation techniques relied on observations from boats. Data obtained in this way however contains bias. Biologging can be used to collect useful data for albatross conservation, such as an overlap of the distribution of albatross and fishing grounds, the fishing boat following behavior of albatross, and the dynamics of wintering areas.
1.Use of biologging
 The study of seabird behavior on the ocean has advanced rapidly thanks to biologging: field research techniques to collect information of the location and behavior of individual animals by attaching a small data recording device known as a “data logger.” Some devices are extremely valuable to scientists because they can track the movement of individuals in time scales of seconds to days, or to an accuracy ranging from 10 m to 200 km. Scientists have learned about food searching behavior, such as seabirds flying in a zigzag pattern when they are looking for food in distinctive oceanographic landscape and features such as continental shelf and slope, current fronts and ocean gyres, or when they are approaching food patches from downwind using odor cues. Observation of the sea behavior of most species of seabird is difficult. Accordingly, based on bio-logging techniques, scientists have been able to collect as yet unrecorded behavioral data related to migration, such as on time spent in the open sea out of breeding season, or how wind affects breeding location and movement between ocean areas out of the breeding season. At the same time, this newly acquired data on behavior at sea is extremely useful for seabird conservation.
The study of seabird behavior on the ocean has advanced rapidly thanks to biologging: field research techniques to collect information of the location and behavior of individual animals by attaching a small data recording device known as a “data logger.” Some devices are extremely valuable to scientists because they can track the movement of individuals in time scales of seconds to days, or to an accuracy ranging from 10 m to 200 km. Scientists have learned about food searching behavior, such as seabirds flying in a zigzag pattern when they are looking for food in distinctive oceanographic landscape and features such as continental shelf and slope, current fronts and ocean gyres, or when they are approaching food patches from downwind using odor cues. Observation of the sea behavior of most species of seabird is difficult. Accordingly, based on bio-logging techniques, scientists have been able to collect as yet unrecorded behavioral data related to migration, such as on time spent in the open sea out of breeding season, or how wind affects breeding location and movement between ocean areas out of the breeding season. At the same time, this newly acquired data on behavior at sea is extremely useful for seabird conservation.
Pelagic seabirds, and the albatross family in particular, show a slow reproduction rate: they start breeding at around 10 years of age; produce one egg at each breeding attempt and breed every other year. On the other hand, the survival rate of parent birds is high at around 95%. For this reason, even a small decrease in the survival rate of parent birds at sea can induce the decrease of the population. There is a risk that fisheries by-catch can lead to a drop in the survival rate of adults. Fifteen of the twenty-two species in the albatross family are on the red list. Therefore, various methods have been developed to reduce the risk of by-catch, such as “tori-lines,” a bird scaring device attached to a boat’s stern, and weighted lines or hooks to submerge the bait quickly. We have learned that if these methods are used appropriately they are highly effective. Yet, ten years after these measures first began to be brought in, the population decline for some albatross species continues, and we do not completely know why.
Although research of fisheries by-catch risk and methods to reduce that risk to date has relied on observation and reports from observers, this data contains bias. In addition, only very few boats carry observers. Using biologging we can obtain information that would be impossible with observation from the boats. Since biologging involves attaching a device to seabirds, there is some effect – whether large or small – on the individual seabird. The information obtained by this technique, however, is extremely important for evaluating risks to the seabird population on the ocean, and for devising conservation strategies. The environmental change and risks related to fishing vary depending on the ocean area. Bearing this in mind, careful use of biologging to obtain low-bias information on behavior in various ocean areas may help us better understand the current state of the albatross family, as well as protect it.
2.Overlap between fisheries and albatross
There is a strong link between bycatch caused by longlines and the reduction in the population of albatross. Evaluation of the impact of fishing and reducing that risk are important issues in albatross conservation. For that reason, it is important to obtain unbiased information on ocean areas with a high risk of bycatch (i.e. areas where longline fishing and the distribution of albatross overlap); and biologging is a powerful tool to evaluate the overlap. For example, there is research that uses geolocators (small recorders that estimate longitude and latitude by recording light levels from sunrise to sunset) to track the annual movements of breeding Laysan albatross and black-footed albatross in the Hawaiian Islands.[i] First, since only some of the individual birds were actually tracked in this research, habitat models were devised to explain the distribution of the two species based on the variations in the ocean environment. Using these models, the distributions of the two species were estimated, and an analysis of the overlap between the estimated distributions of the two species and longline fishing areas in Hawaii were carried out. As a result, it was found that while black-footed albatross often use the areas where longline bycatch occurs, the Laysan albatross does not. In other words, they surmised that the bycatch risk was higher for black-footed albatrosses. Despite the fact that there are ten times fewer breeding black-footed albatrosses as Laysan in this region, black-footed albatrosses suffer bycatch more frequently, and it seems that the bycatch risk is related to overlap between fisheries and albatross distribution.
It is not always the case, however, that the bycatch risk is high in the regions that have both a high density of albatross and intensive fishing (measured by hooks per area unit). Apart from the number of albatross in the vicinity of the fishing boats, various factors affect the bycatch rate (bycatch per 1,000 hooks), such as: climate and features of the fishing boats, or devices used to reduce bycatch. For example, albatross bycatch rate due to longline fishing in Hawaii is affected by season and oceanic environments such as climate indices related to El Niño, the timing of bycatch reduction measures, the way tori lines are used, and bait dyeing[ii]. It has also been reported that specific boats have a high bycatch rate[iii]. In addition, as well as these external factors, it may be that the likelihood of albatross bycatch is affected by variation related to sex and the individual. Meanwhile, not all bycatch is reported; an issue is that information from poaching vessels is not available.
3.Albatross follow fishing vessels
GPS data loggers (which are much more accurate that geolocators) are especially useful to study how seabird individuals follow fishing boats. This GPS tracking shows individual variation in seabird vessel-following behavior causes a latent bycatch risk. In Wales, UK, the movement data was collected on breeding northern gannets using GPS loggers, and the movements of fishing boats was simultaneously recorded using the GPS location recording Vessel Monitoring System (VMS). It was found that some individuals fed near fishing boats, and some did not, and that this trend continued for several weeks[iv]. On the other hand, black-browed albatrosses on the Patagonian Shelf were tracked using GPS loggers while VMS was used to know the movement of fishing trawlers. Although it was confirmed that 17% of the individuals followed fishing boats, no individuals were found to consistently follow boats[v]. It depends on the situation, but the frequency of vessel-following behavior may vary according to sex and the individual. In situations where there is this kind of individual and sex variation associated with bycatch risk, we can investigate it, then make a good evaluation of its effects by analyzing the relationship between bycatch risk and variation among individuals in a population.
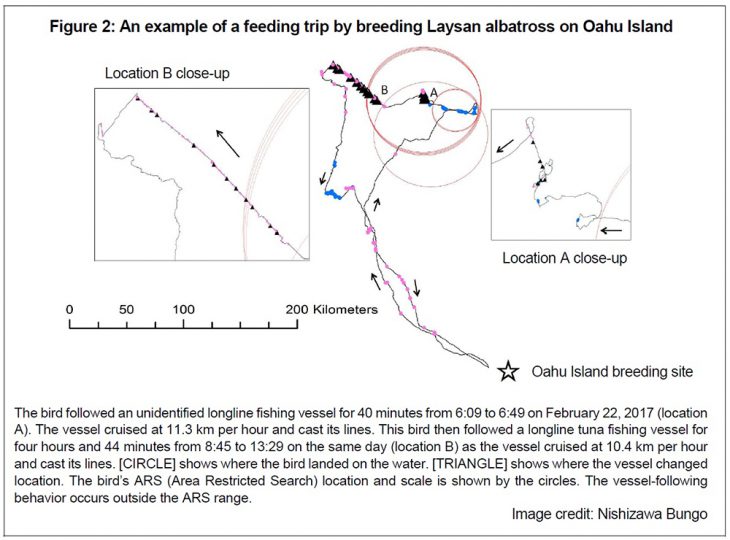
By using image data collected by video and still picture loggers attached to albatrosses, we can learn a lot. The author and colleagues attached GPS and camera loggers to incubating parent birds of Laysan albatross breeding at Oahu Island. Both recorded their movements and image data at the same time (figure 1). As a result, pictures were clear enough to identify boat names, and it was ascertained that birds follow longline-fishing boats when the boats raised and cast long-lines. Laysan albatross suddenly changed their direction when they were 10 km away from the longline-fishing boats, then approached the boats and followed them for around two hours. They apparently discover fishing boats that are just under 10 km away (figure 2).
With the position and image data, we can analyze the range from which albatross approach fishing vessels and their behavior in the ships’ vicinity. The information of the range within which albatross are attracted to longline fishing vessels can be used for estimating risk. These image data may also be useful for estimating small-scale by-catch risk, particularly in sea areas where VMS systems have not been introduced, and where poaching vessels are numerous.
4.Genetic groups and the areas they use during the non-breeding season
We now know that seabirds of the procellariiforms spend the non-breeding season in the sea thousands of kilometers away from their breeding sites. These non-breeding regions are often far away from the countries where their breeding sites are located. The marine environment and fishing-associated risks in these non-breeding regions are very different from those in the breeding regions. It is not easy to collect information from these offshore regions. Making the problem even more complicated is that, even in breeding groups that belong to the same species, there are multiple groups that are genetically different, and these groups sometimes spend the non-breeding season in different regions. These “hidden” genetic groups should be treated separately when these groups are under the issue of conservation. Meanwhile, we need to study non-breeding regions for each different genetic group, and understand the impact of climate change and fishing in these non-breeding season regions.
Figure 3: An albatross spending the non-breeding
season on the Bering Sea

Photo credit: Nishizawa Bungo
Using mitochondrial DNA analysis, we have recently learned that even albatross (figure 3) that breed in Japan are genetically divided into Torishima and Senkaku lineages[vi]. It has been reported that albatrosses breeding at Torishima spend the non-breeding season in the vicinity of the Aleutian Islands and continental shelf slope area of the Bering Sea[vii]. It is also known, however, that after leaving the colony some young birds visit the Sea of Okhotsk during their first year. For that reason, it is necessary to investigate whether these two lineages spend the non-breeding season in different sea regions, and if so whether the risks in those areas differ.
The risks out at sea are not limited to fishing and lack of food. Ocean pollution is a problem; egg shells of seabirds became thinner owing to persistent organic pollutants (DDE), and large numbers of birds died in the 1970s. Biologging provides information about the areas of high seas that have never been surveyed before, and tells us that ocean pollution varies greatly depending on the sea region. For example, some short-tailed shearwater individuals that breed in Tasmania spend the non-breeding season in the southeast of the Bering Sea, while others in the Sea of Okhotsk. We have learned that the individuals that spend time in the Sea of Okhotsk showed eight times higher concentrations of mercury in their flight feathers than those visiting the south east Bering Sea[viii]. Individuals that use a different sea region during the non-breeding season face a different risk from pollution. When the regions used during the non-breeding season are different, and when the pollution and bycatch risks in those regions are different, it may become necessary to implement specific measures to deal with the different risks facing various conservation units. The application of tracking to investigate regions where birds spend the non-breeding season is likely to be of broad use when devising seabird conservation strategies.
5.Seabird tracking and marine IBAs
The high sea is affected by human activities such as shipping traffic, ocean pollution and fishing activity. One method of protecting seabirds from such human activity is to set up marine reserves and to manage them in some way. As of 2016, Bird Life International, a global NGO set up to protect seabird species, had identified 3,300 Marine IBAs (Marine Important Bird and Biodiversity Areas). Birds of the procellariiforms (including albatross species) live in the high seas both during the breeding season and the non-breeding season, so it is difficult to observe them. Tracking is essential in order to set up Marine IBAs for their protection.
By tracking many individuals of multiple species over the course of a year, the regions necessary for the protection of southern hemisphere albatross have been identified. For example, in order to set up an ocean-scale IBA from the Indian Ocean to Southern Atlantic, research has used tracking data of multiple individuals (during the breeding and non-breeding seasons) belonging to ten species of seabird, including the Amsterdam albatross (critically endangered), the wandering albatross (vulnerable), and Indian yellow-nosed albatross (threatened)[ix]. As a result, it has become known that only 1.8% of areas intensively used by seabirds were included in existing MPAs set up in exclusive economic zones, and 63% were in international waters. Likewise, in the Indian Ocean, tracking data of threatened seabird species breeding on the Tristan da Cunha island group showed that the areas intensively used by seabirds were all in the high sea, and that none were in existing South Atlantic MPAs[x]. In this way, the Marine IBAs for offshore living seabirds (albatrosses are the main subject) are actually in international sea areas where it is difficult to set up. At present, the only conservation tool we have is fisheries management via regional fisheries management organizations such as the International Commission for the Conservation of Atlantic Tunas (ICCAT). These recent advances in the analysis of tracking data once again demonstrate the need for a new multinational approach to conservation of such highly-mobile open sea species.
Tracking data of seabirds tells us about more than just important locations for the conservation of seabirds. Seabirds themselves search areas where food organisms are abundant and consume organisms of various trophic levels such as squid, zooplankton, and sardines and other pelagic fish that swim in schools. The distribution of seabirds responds to the distribution of these organisms, as well as rapid changes of their distributions[xi]. In addition, seabirds have a high per-individual energy consumption, so it is thought that the areas where seabirds gather are indications of large energy flows through multiple trophic levels, from phytoplankton and zooplankton to fish and higher-order predators[xii][xiii]. Accordingly, Marine IBAs set up using seabird tracking data may also be useful for establishing EBSAs (Ecologically or Biologically Significant Areas) selected for “large energy flows through highly-productive levels of the food chain” or “high diversity.”
6.Conclusion
The term biologging originated in Japan, and researchers here have used this technique to conduct outstanding research on seabirds, marine mammals, sea turtles, and large fish behavior and physiology. As I have described in this paper, research of seabirds using this technology is taking place all over the world, and the data collected in Japan to date, and in the future, is expected to be of great use for the conservation of northern hemisphere albatross species. What’s more, the work of Japanese researchers, particularly internationally important research using accelerometer and visual loggers, is expected to be of use to seabird protection and the management of marine ecosystems.
[Acknowledgements]
Comments were received from Dr. Tomohiro Deguchi (see “The Conservation of Endangered Albatross Species”) during the preparation of this text, while photos and figures were kindly lent by Nishizawa Bungo.
Related article:
The Conservation of Endangered Albatross Species
https://www.japanpolicyforum.jp/archives/science/pt20181113113535.html
Translated from “Tokushu II Sekai no Ahodori no hogo to hozen / 2 Ahodori no hozen no aratana tenkai (Special Feature II The Preservation and the conservation of the world’s albatross / 2 New Developments in Albatross Conservation—Using biologging to elucidate behavior in the ocean),” Seibutsu No Kagaku Iden, Vol. 72 No. 2, February 2018, pp. 1654-170. (Courtesy of NTS Inc.) [February 2018]
[i] Zydelis, R., Lewison, R. L., Shaffer, S. A., Moore, J. E., Boustany, A.M. et al. Proc.R Soc. Lond. 278, 3191-3200 (2011).
[ii] Gilman, E., Chaloupka, M., Peschon, J. & Ellgen, S. PLoSONE 11, e0155477. doi:10.1371/journal.pone. 0155477 (2016).
[iii] Dietrich, K. S., Parrish, J. K. & Melvin, E. F. Biol Conserv. 42, 2642-2656 (2009).
[iv] Patrick, S. C., Bearhop, S., Bodey, T. W., Grecian, W. J., Hamer, K.C. et al. J. Avian Biol. 46, 431-440 doi: 10.1111/jav.00660 (2015).
[v] Granadeiro, J. P., Brickle, P. & Catry, P. Anim. Conserv. 17, 19-26 (2014).
[vi] Eda, M., Higuchi, H. et al. Japanese Journal of Ornithology. 61, 263-272 (2012).
[vii] Suryan, R. M., Dietrich, K. S., Melvin, E. F., Balogh, G. R., Sato, F. et al. Biol. Conserv. 137, 450-460 (2007).
[viii] Watanuki, Y., Yamamoto, T., Yamashita, A., Ishii, C., Ikenaka, Y. et al. J. Ornithol. 156, 847-850 (2015).
[ix] Delord, K., Barbraud, C., Bost, C.-A., Deceuninck, B., Lefebvre, T. et al. Marine Policy 48, 1-13 (2014).
[x] Dias, M. P., Oppel, S., Bond, A. L., Carneiro, A. P. B., Cuthbert, R. J. et al. Biological Conservation 211, 76-84 (2017).
[xi] Piatt, J. F., Sydeman, W. J. & Wiese, F. Mar. Ecol. Prog. Ser. 352, 199-204 (2007).
[xii] Sydeman, W. J., Brodeur, R. D., Grimesj C. B., Bychkov, A. S. & McKinnell, S. Deep-Sea Res II 53, 247-249 (2006).
[xiii] Hyrenbach, K. D., Forney, K. A. & Dayton, P.K. Aquat. Conserv. Mar and Fresh Ecosys. 10, 437-458 (2000).
Keywords
- albatross
- biologging
- GPS data logger
- Hawaiian Islands
- bycatch rate
- by-catch risk
- longline-fishing boat
- ocean pollution
- Torishima Island
- Sea of Okhotsk
- Aleutian Islands
- Bering Sea
- Bird Life International
- Marine Important Bird and Biodiversity Areas
- Ecologically or Biologically Significant Areas